CLEAR-AI Study at a Glance
Consensus on Ethical Use of AI in Medical Manuscript Writing
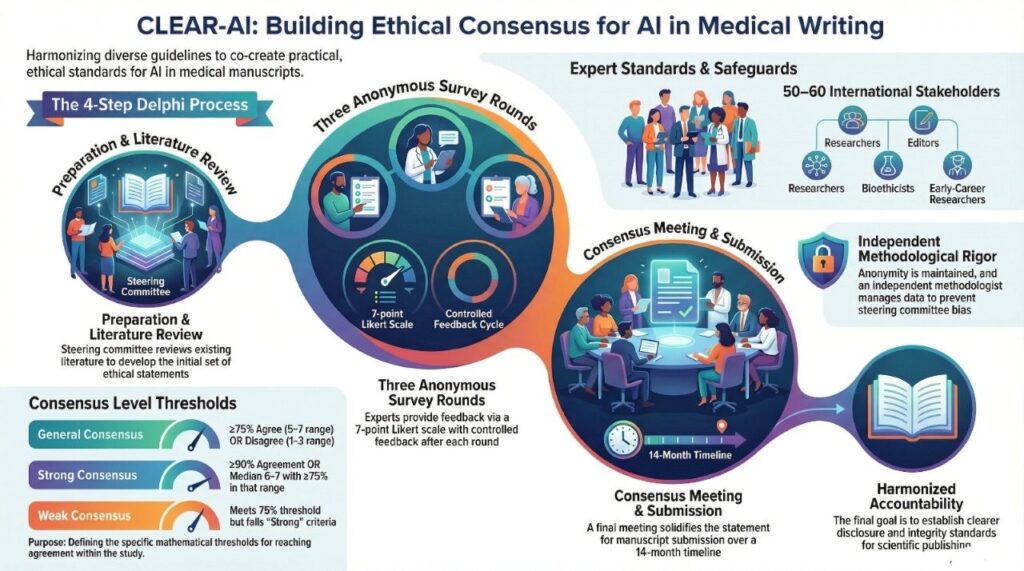
Delphi Study Protocol
Steering Committee
| Name | Role |
| Saurabh Kumar Das | Editor, Clinician |
| Ravi Jain | Clinician, Reviewer, Delphi Methodologist |
| Prashant Nasa | Editor, Clinician, Delphi Methodologist |
| Yash Mathur | AI Engineer |
| Avneesh Khare | Clinician, AI Expert |
| Open to recruit | Additional members welcome |
Objective
Develop a multi‑stakeholder consensus statement on the ethical use of generative AI in medical writing, using a modified Delphi process.
Why Now?
- AI tools (ChatGPT, Gemini, etc.) are transforming medical writing.
- Existing guidelines vary widely across journals → confusion for authors.
- Need for harmonized, practical guidance co‑created by diverse experts.
Expert Panel
- 50–60 international experts
- Diverse stakeholders: researchers, editors, medical writers, bioethicists, statisticians, publishers, early‑career researchers
- Balanced geographical representation
Delphi Process
| Step | Description |
| 1 | Steering committee + literature review → initial statements |
| 2 | 3 anonymous survey rounds (7‑point Likert scale) |
| 3 | Controlled feedback after each round |
| 4 | Consensus meeting + final statement |
Consensus Definition
- Consensus achieved: ≥75% agreement (score 5–7) or disagreement (1–3)
- Strong consensus: ≥90% agreement or median 6–7 (agree) / 1–2 (disagree) with ≥75% in that range
- Weak consensus: meets 75% but not strong criteria
Key Safeguards
- Anonymity among panelists
- Independent Delphi methodologist manages surveys & data
- No voting by steering committee members
- Attrition & stability monitored
Timeline
~14 months – from literature review to manuscript submission
Expected Impact
✔ Harmonized guidance for authors, editors, and institutions
✔ Clearer disclosure & accountability standards
✔ Supports integrity of scientific publishing in the AI era
Steering committee open to recruiting additional members.
Contact information available upon request.